- Home
- Heterogeneous catalysis
Heterogeneous catalysis
Heterogeneous catalysis is one of the cornerstones of chemical industry, and more efficient catalysts are key in making the world more energy efficient and less polluting. The drive for sustainable energy solutions requires additional focus on catalysis as it is essential to be able to efficiently transform energy from one chemical form into another. In many sustainable energy technologies, the lack of efficient and economically viable catalysts is the primary factor limiting their use.
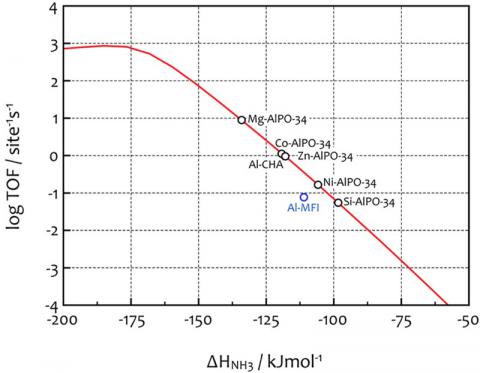
SUNCAT research in thermochemical transformations is focused on the conversion of new feedstocks into base chemicals and transportation fuels as well as chemical transformations related to environmental catalysis. Our portfolio includes hydrogenation of CO and CO2, transformation of methanol to synfuels, decomposition of NO and oxidation of CO. An understanding of how catalysts work at the atomic-scale can be obtained from density functional theory calculations. If these calculations are coupled with (micro-)kinetic models of surface processes an in-depth understanding of the factors that limit the catalysts performance can be achieved. This often enables the prediction of trends among different catalytic materials (see Figure). At SUNCAT, our research is focused on transition-metal based catalysts, various oxide materials as well as zeolites and zeotypes.
