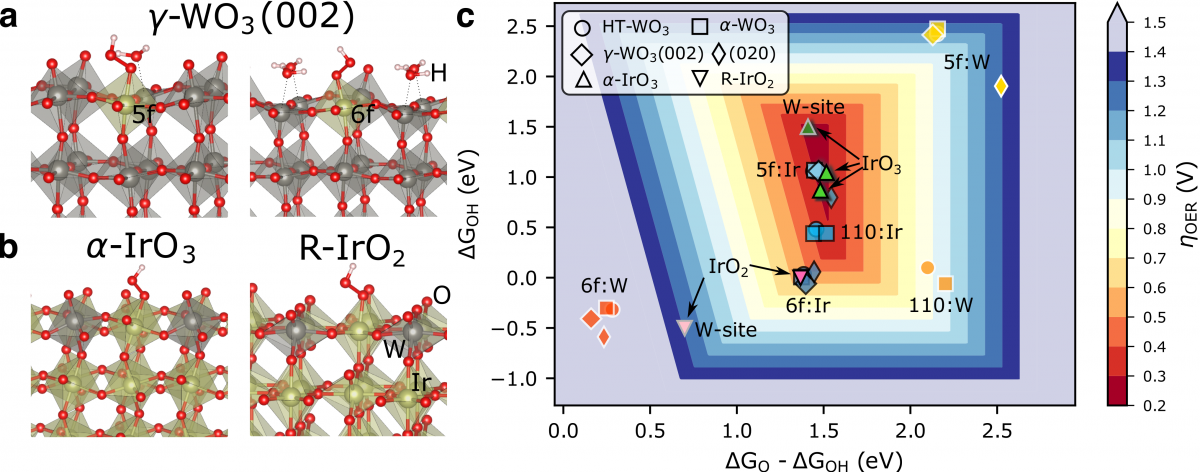
Electrolysis coupled with renewable energy is essential for green hydrogen production and its application to achieving deep decarbonization. The efficiency of electrolysis is limited by the oxygen evolution reaction (OER), which requires an efficient, low-cost, and acid-stable catalyst for proton exchange membrane (PEM) electrolysis. In this work, we designed a highly active, acid-stable catalyst involving iridium-tungsten trioxide (Ir:WO3/Ir). Our theory predicts that high-valence Ir atoms are active sites for the OER and they can be stabilized in the WO3 matrix. Experimentally, we showed that the Ir:WO3/Ir catalyst achieves a high mass activity of 13.8 A mgIr–1 at 10 mA cm–2 and a stability in 0.5 M H2SO4 for 32 days; such a performance originates from the stabilized high-valence Ir sites with optimized binding energies to oxygen species. Our work demonstrates an effective strategy to obtain low-cost, durable, and efficient OER catalysts to potentially broaden the adoption of PEM electrolyzers. Computational Data are available at https://www.catalysis-hub.org/publications/ShiLong-term2021.